|
Q.No.
|
Question
|
Marks
|
|
1.
|
Define electron gain enthalpy.
|
1
|
|
2.
|
Write I.U.P.A.C. name of the following compound:

|
1
|
|
3.
|
Define standad enthalpy of formation.
|
1
|
|
4.
|
Write electronic configuration of $mathrm{Cu}^{2+}$ ion. (Atomic number of $mathrm{Cu}=29$ ).
|
1
|
|
5.
|
In which orbital will the electrons enter first 3 d or 4p ?
|
1
|
|
6.
|
Give the units of vander waal’s constants. Also point out their significance.
|
2
|
|
7.
|
$0.3780 mathrm{~g}$ of an organic chloro compound gave $0.5740 mathrm{~
g}$ of silver chloride in carius estimation. Calculate the percentage of chlorine present in compound.
|
2
|
|
8.
|
Write the short notes on:
|
2
|
|
(a)
|
Wurtz Reaction
|
|
|
(b)
|
Freidal Craft’s Alkylation
|
|
|
9.
|
Write the molecular shapes of:
|
2
|
|
(a)
|
$mathrm{XeF}_{4}$
|
|
|
(b)
|
$mathrm{CIF}_{3}$
|
|
|
10.
|
Determine the emperical formula of an oxide of iron which has $69.9 %$ iron and $30.1 %$ dioxygen by man. $quad$ [Atomic mass: $mathrm{Fe}=55.85, mathrm{O}=16.00]$
|
2
|
|
11.
|
The ionization enthalpy of lithium is $520 mathrm{kJmol}^{-1}$, calculate the amount of energy required to convert $140 mathrm{mg}$ of lithium atoms in gaseous state into $mathrm{Li}^{+}$ ion.
|
2
|
|
12.
|
How does Heisenberg’s uncertanity principle support concept of orbital?
|
2
|
|
13.
|
Compelete the following reactions:
|
3
|
|
(a)
|

|
|
|
(b)
|
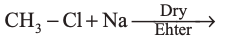
|
|
|
(c)
|

|
|
|
14.
|
Balance the following reaction in acidic medium:
$mathrm{MnO}_{4}^{-}(mathrm{aq})+mathrm{SO}_{2}(mathrm{~g}) longrightarrow mathrm{Mn}^{2+}(mathrm{aq})+mathrm{HSO}_{4}^{-}(mathrm{aq})$
|
3
|
|
15.
|
The value of $mathrm{K}_{mathrm{C}}$ for the reaction:
$2 mathrm{~A} rightleftharpoons mathrm{B}+mathrm{C}$ is $2 times 10^{-3}$ at $500 mathrm{~K}$. At given time, the composition of reaction mixture is $[mathrm{A}]=[mathrm{B}]=[mathrm{C}]=3 times 10^{-4} mathrm{M}$. Is the reaction mixture at equilibrium? If not, what is the direction of net reaction?
|
3
|
|
16.
|
Write down the nature of below reaction with reason:
|
3
|
|
(a)
|
$mathrm{NH}_{3}+mathrm{H}^{+} rightarrow mathrm{NH}_{4}^{+}$
|
|
|
(b)
|
$mathrm{BF}_{3}+mathrm{NH}_{3} rightarrow mathrm{F}_{3} mathrm{~B} leftarrow mathrm{NH}_{3}$
|
|
|
(c)
|
$mathrm{H}_{2} mathrm{SO}_{4} rightarrow mathrm{H}^{+}+mathrm{HSO}_{4}^{-}$
|
|
|
17.
|
Arrange the following carbocation in creasing order of their stability. $left(mathrm{CH}_{3}right)_{2} mathrm{CH}^{+}, mathrm{CH}_{3} mathrm{CH}_{2}^{+},left(mathrm{CH}_{3}right)_{3} mathrm{C}^{+}, mathrm{CH}_{3}^{+}$
|
3
|
|
18.
|
How will you convert
|
3
|
|
(a)
|
Propan-1-ol into propene
|
|
|
(b)
|
2-bromopropane into But-2-ene
|
|
|
(c)
|
Ethyl amine into ethyl isocyanide
|
|
|
19.
|
|
3
|
|
(a)
|
Define eutrophication and penumocanosis.
|
|
|
(b)
|
Write difference in between photochemical and classical smog.
|
|
|
20.
|
|
3
|
|
(a)
|
Calculate the oxidation number of $mathrm{S}$ in $mathrm{S}_{2} mathrm{O}_{6}^{-2}$ having $(-mathrm{O}-mathrm{O}-)^{2-}$ linkage and $mathrm{C}$ in $mathrm{CH}_{3} mathrm{COOH}$.
|
|
|
(b)
|
Balance the equation in basic medium by half reaction method
$$
mathbf{P}_{4}(s) rightarrow P H_{3}(g)+H_{2} P O_{2}^{-}(A q)
$$
|
|
|
21.
|
|
3
|
|
(a)
|
Out of staggerd and eclipsed conformations of n-butane, which is more stable and why?
|
|
|
(b)
|
What causes the temporary and permanent hardness of water.
|
|
|
22.
|
Write a breif note on the following environmental terms:
|
3
|
|
(a)
|
Acid rains
|
|
|
(b)
|
Eutrophication/Green House effect
|
|
|
(c)
|
Green chemistry
|
|
|
23.
|
|
3
|
|
(a)
|
Define buffer solution.
|
|
|
(b)
|
The solubility of $mathrm{Sr}(mathrm{OH})_{2}$ at $298 mathrm{~K}$ is $19.23 mathrm{~g} / mathrm{L}$ of solution. Calculate the concentration of strontium and hydroxyl ions and the $mathrm{pH}$ of the solution.
|
|
|
24.
|
Write any three main biological importance of $mathrm{Ca}$ and $mathrm{Mg}$.
|
3
|
|
25.
|
|
5
|
|
(a)
|
The stability of peroxide and superoxide of alkali metals increases as we go down the group. Explain giving reasons.
|
|
|
(b)
|
How to control photochemical smog.
|
|
|
OR
|
|
|
(a)
|
Derive first law of thermodynamics.
|
|
|
(b)
|
Define enthalpy of neutralisation.
|
|
|
(c)
|
Calculate the $Delta mathrm{H}^{theta}$ of the reaction.

Bone enthalpies of $mathrm{C}-mathrm{H} & mathrm{C}-mathrm{Cl}$ bond are $415 mathrm{~kJ} mathrm{~mol}^{-1} & 326 mathrm{~kJ} mathrm{~mol}^{-1}$
|
|
|
26.
|
|
5
|
|
(a)
|
Account for the following:
|
|
|
i.
|
Boron Halides do not dimerise like $mathrm{BH}_{3}$.
|
|
|
ii.
|
Carbon shows catenation
|
|
|
iii.
|
$mathrm{PbCl}_{4}$ is a good oxidising agent.
|
|
|
(b)
|
Complete the following reactions:
|
|
|
i.
|
$mathrm{B}_{2} mathrm{H}_{6}+3 mathrm{O}_{2} longrightarrow$
|
|
|
ii.
|
$2 mathrm{BF}_{3}+6 mathrm{NaH} stackrel{450 mathrm{~K}}{longrightarrow}$
|
|
|
OR
|
|
|
(a)
|
Write equation to justify amphoteric nature of Water.
|
|
|
(b)
|
What is application of equilibrium constant.
|
|
|
(c)
|
What are the full form of BOD?
|
|
|
27.
|
|
5
|
|
(a)
|
Is the entropy of the universe constant?
|
|
|
(b)
|
If standard free energy change for a reaction is found to be zero, what is its equilibrium constant.
|
|
|
(c)
|
Define common ion effect.
|
|
|
OR
|
|
|
(a)
|
Calculate the degree of ionisation of $0.1 mathrm{~mol} / mathrm{L}$ solution of acetic acid, given $mathrm{K}_{mathrm{a}}$ for $mathrm{CH}_{3} mathrm{COOH} 1.8 times 10^{-5} mathrm{~mol} / mathrm{L}$
|
|
|
(b)
|
Define the following terms :
|
|
|
i.
|
Solubility product
|
|
|
ii.
|
Buffer solution
|
|
|
iii.
|
Henderson equation
|
|


